
The hole bored in the top plate was very small.ģ. The two plates were 16 mm across, "correct to about. 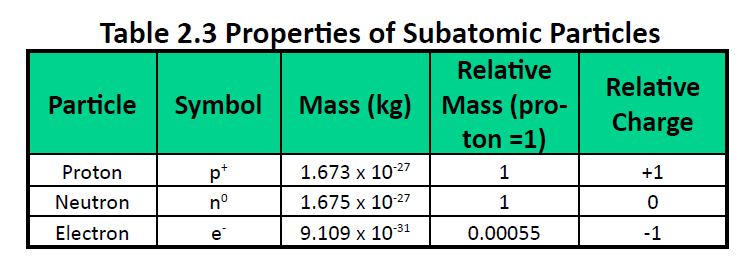
These are some points to be made about the experiment:ġ. This is a photo dating from the time of the experiment. Through the third of these windows, set at an angle of about 18° from the line Xpa and in the same horizontal plane, the oil drop is observed. To the three windows g (two only are shown) in the brass vessel D correspond, of course, three windows in the ebonite strip c which encircles the condenser plates M and N. The air about the drop p was ionized when desired by means of Röntgen rays from X which readily passed through the glass window g. The atomizer A was blown by means of a puff of carefully dried and dust-free air introduced through the cock e. A long search for causes of slight irregularity revealed nothing so important as this and after the bath was installed all of the irregularities vanished. This constant temperature bath was found essential if such consistency of measurement as is shown below was to be obtained. long, and a cupric chloride cell d, and second by immersing the whole vessel D in a constant temperature bath G of gas-engine oil (40 liters) which permitted, in general, fluctuations of not more than. Complete stagnancy of the air between the condenser plates M and N was attained first by absorbing all of the heat rays from the arc A by means of a water cell w, 80 cm. 
down these were measured with a very carefully made mercury manometer M which at atmospheric pressure gave precisely the same reading as a standard barometer. The brass vessel D was built for work at all pressures up to 15 atmospheres but since the present observations have to do only with pressures from 76 cm. The experimental arrangements are shown in Fig. Here is a diagram of his apparatus, reproduced from his 1913 article: His 1913 article announcing the determination of the electron's charge is a classic and Millikan received the Nobel Prize for his efforts. Millikan started his work on electron charge in 1906 and continued for seven years. This work continued until about 1901 or 1902. Thomson, Townsend, and Wilson each obtained roughly the same value for the charge on positive and negative ions. He determined the e/m ratio of the droplets (2 divided by 4), then multiplied by the mass of one droplet to get the value for e. the total mass of all water droplets (found by measuring the acid's increase in weight) the total electric charge carried on all the droplets (this was done by absorbing the water into an acid and measuring the charge picked up.)Ĥ. the mass of a water droplet (actually the average mass of many)Ģ. Several items were measured in this experiment.ġ. Under the influence of gravity, the drop would fall, accelerating until it hit a constant speed. Townsend's work depended on the fact that drops of water will grow around ions in humid air. Townsend's work will be described as an example. The charge on electron was first measured by J.J. Determination of the Charge on an Electron Determination of the Charge on an Electron Each such orbital can be occupied by a maximum of two electrons, each with its own projection of spin m s. As an alternative to the magnetic quantum number, the orbitals are often labeled by the associated harmonic polynomials (e.g., xy, x 2 − y 2). Įach orbital in an atom is characterized by a set of values of the three quantum numbers n, ℓ, and m l, which respectively correspond to the electron's energy, its angular momentum, and an angular momentum vector component ( magnetic quantum number). The term atomic orbital may also refer to the physical region or space where the electron can be calculated to be present, as predicted by the particular mathematical form of the orbital. This function can be used to calculate the probability of finding any electron of an atom in any specific region around the atom's nucleus. 
In atomic theory and quantum mechanics, an atomic orbital ( / ˈ ɔːr b ɪ t ə l/) is a function describing the location and wave-like behavior of an electron in an atom. To see the elongated shape of ψ( x, y, z) 2 functions that show probability density more directly, see pictures of d-orbitals below. Each picture is domain coloring of a ψ( x, y, z) function which depends on the coordinates of one electron. The two colors show the phase or sign of the wave function in each region. The shapes of the first five atomic orbitals are: 1s, 2s, 2p x, 2p y, and 2p z. For the collection of spaceflight orbits, see Orbital shell (spaceflight).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
